-
Introduction to Graphene
Jun 01, 2017 | ACS MATERIAL LLCGraphene is defined as a single-atomic-layer, 2-dimensional allotrope of carbon atom. This material has superlative properties of strength, elasticity, electrical conductivity, and thermal conductivity. It can potentially provide a higher energy density than that of a common supercapacitor. There are multiple ways to prepare graphene using one of the top-down or bottom-up categorical methods but the most popular within these categories would be the chemical vapor deposition (CVD) as it can produce high-quality graphene film which can be transferred onto a desired substrate relatively easily. The cost efficiency of graphene, along with its spectacular properties, proves to be useful in many applications, including electronics, solar cells, sensors, structural reinforcements, and almost unlimited other possibilities. Its vast popularity in the research and technical fields indicates that graphene may be the next big thing.
Introduction to Graphene
Graphene is a two-dimensional, crystalline allotrope of carbon atoms that form a hexagonal lattice structure with an sp2 hybridization. This 96.6% optically transparent material is the strongest known to mankind per unit size with incredible electrical properties and the best heat conductor at room temperature which can lead to many applicable uses in the research and technical industries. With a molecular bond length of 0.142 nanometers and an atomic thickness of 0.345 nanometers, thermal instability is prevented due to its carbon-to-carbon bonds that are so small yet strong. Its tensile strength is far greater than steel at approximately 0.4 GPa while having elastic properties with Young’s Modulus of 500 GPa, much more elastic than rubber.
Preparation Methods
Graphene can be prepared with a “top-down” approach that involves obtaining a single-layer graphene isolated from multilayer graphite crystals, or with a “bottom-up” method causing the graphene crystals to self-assemble from small molecules used to create graphene, respectively. One top-down method incorporates mechanical peeling using the “Scotch tape method” to mechanically peel pyrolytic graphite crystals, creating a single-layer graphene material. While this method is too inefficient to create large quantities, it represents the method of the original invention (2004, University of Manchester, UK) and provided sufficient material to demonstrate many of the superlative properties that are now being thoroughly exploited.
Another top-down method involves the liquid-phase exfoliation, a different mechanical process used to get a single-layer graphene and is more advantageous as large quantities can be produced. Delamination requires exfoliating agents (solvents or molecules) to disperse graphene using the mechanical force needed to prevent the overlapping between adjacent layers within the graphite crystals.1 A stabilizing agent is then used to prevent restacking of the crystal and maintain stable graphene flakes. Then, either through shear/turbulence mixing, ball milling, ultrasonication or electrochemical potential, exfoliation can be achieved.
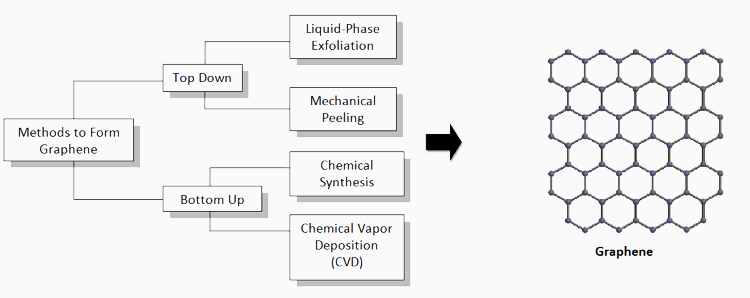
Figure 1. The flow chart shows the various methods to create or obtain graphene from graphite crystals or molecules.
The bottom-up approach provides a more structural and molecular technique of creating graphene that can be acquired with the possibility of chemical synthesis. Through this method, small-sized nanographene or graphene nanoribbons (GNRs) are formed with the smallest nanographene size ranging from 1 to 5 nanometers and the largest synthesized monodispursed nanographene molecule containing more than 200 carbon atoms with a disk diameter of 3.2 nanometers.2,3 GNRs are formed with the reaction of high aromatic rings and polycyclic aromatic hydrocarbons. The issue with compounds having greater than 200 carbon atoms is that they are hardly soluble in organic solvents needed to process materials for device applications. The way to counteract this hurdle is to add alkyl chains to the outer portions of the molecule in order to form a highly ordered superstructure with its phase-forming behavior.4 Graphene molecules can then arrange itself in solution with its proper substituents.
However, the most current and trending method to create graphene from the bottom-up is through chemical vapor deposition (CVD) which has an easy laboratory set-up and already been established in an industrial setting. Treating the substrate before beginning the CVD process is commonly done to improve graphene formation and obtain a smoother surface. Wet chemical treatments are considered for copper films to prevent oxide reduction and if this step is taken, the copper film must be soaked in acetic acid.5 Then, through CVD, a metal substrate, typically copper, is heated in a reaction chamber under low vacuum.
Decomposed methane supplies the carbon needed to form graphene and the gaseous reactants from methane and hydrogen gases inside the chamber set at high temperatures are deposited onto the substrate where a reaction will occur on the substrate’s surface. This is caused when hydrogen is catalyzing a reaction between the methane and the metal substrate. Carbon atoms from the methane are then deposited onto the substrate surface through chemical absorption. Some factors that affects the outcome of the graphene are cooling rate, carbon exposure time and its concentration, flow rate and the supply of carbons.6
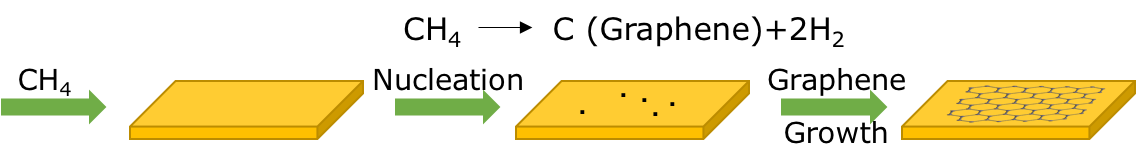
Figure 2. The chemical vapor deposition (CVD) process includes the decomposition of methane to use the carbon needed in order to grow on the substrate surface.
Nevertheless, the greatest advantage of CVD is that it allows the graphene to be transferred onto another substrate. Polymethylmethacrylate (PMMA) is often used as a stiffening polymer surface to make this graphene transfer possible and the process of etching out the original substrate can begin. Etching allows for the graphene to remain beneath the polymer so that the graphene layer is protected as it is being removed from the substrate. The PMMA left behind on the graphene layer can be removed with a solvent, leaving the graphene on the substrate of choice, bonded by Van der Waals forces.
Figure 3. a) Trivial Transfer® Graphene b) ACS Material Single Layer Graphene Oxide.
The ACS Material LLC offers CVD graphene on a number of wafer substrates such as copper (Cu), silicon (Si), silicon dioxide (SiO2), quartz, and PET. Customization of the graphene before transfer is also possible. Proper treatment and simple steps can remove PMMA, leaving the graphene on a desired substrate. The patented Trivial Transfer® Graphene has eliminated the tedious step of etching so that it is ready for use.
Applications
With its high elasticity and mechanical strength, graphene provides many uses ranging from electronics to batteries. The market for nanomaterial products is on the rise due to its unique properties as its virtual transparency makes it ideal for touch screens and its high conductivity is great for supercapacitor usage. Many products have been improved, or have a high potential, due to graphene and new findings are being made as better alternatives to current products used today.
Graphene’s small size can allow for multiple uses in electronics and can potentially outperform current silicon-based products. The semimetal material has an electron mobility found to be 200,000 cm2/V∙s which is 6 times higher than copper and can assist in increasing speed higher than 30 PHz of microprocessor speeds.7 Graphene has the highest intrinsic mobility at room temperature which allows a greater switching speed in electronics.8
Graphene is playing an increasing role in energy storage. While supercapacitors can store anywhere from 10 to 100 times more energy and can release that energy fairly quickly, the issue with supercapacitors is the cost. Graphene is cost efficient and has multiple properties that can be exploited to give it great potential as a component of energy storage systems. The material can charge and discharge energy much more quickly and potentially store more energy than a lithium-ion battery. ACS Material uses different methods in providing graphene oxide (with an application field in supercapacitors) which includes Modified Hummer’s Method (H method), Staudenmaier Method (S method) and so on. In the images below, the Modified H Method uses pure graphite powder whereas the S Method incorporates sulfuric acid in combination with nitric acid (HNO3) and potassium chlorate (KClO3).
Batteries are enhanced by graphene due to its lightweight nature and the ability to store high capacity energy. Compared to other batteries, graphene adds conductivity and has a higher surface area with better chemical tolerance and energy density; now, the durability and charge capacity has been improved on lithium-ion battery anodes. The theoretical capacity is limited to 372 (mA∙h)/g but new devices will need more energy capacity. This material enhances lithium-ion batteries so that the process of inserting and extracting Li+ ions is faster, easier and can be reversed.8 The fact that charging times can be shortened and are sustainable for high capacity energy storage puts graphene in the forefront as one of the most miraculous materials known on earth.
Graphene’s characteristics make this one of the most promising of materials to pique the interest this century. Companies and university research laboratories have spent tremendous time and money into making this material even more adaptable for everyday use: from electrical devices to batteries, contact lenses to structural support for the preservation of antiquities. The continued research and discovery cycles for investigation into graphene in both theoretical and practical domains are unabated. There is little doubt that this wonder material will find its way into many of the future marvels of science and society.
ACS Material Products:
- Graphene Series: Single Layer Graphene, Mechanically Exfoliated Single Crystal Graphene, others.
- CVD Graphene: Trivial Transfer® Graphene, Graphene on Cu Foil, Graphene on Si Substrate, others.
References
1. Kumar, Challa Vijaya, and Ajith Pattammattel. Introduction to Graphene: Chemical and Biochemical Applications. Elsevier, 2017.
2. Chen, Long, et al. “ChemInform Abstract: From Nanographene and Graphene Nanoribbons to Graphene Sheets: Chemical Synthesis.” ChemInform, vol. 44, no. 2, Aug. 2013, doi:10.1002/chin.201302190.
3. Guldi, Dirk M., and Nazario Martin. Carbon nanotubes and related structures synthesis, characterization, functionalization, and applications. Weinheim, Wiley-VCH, 2010.
4. Pisula, Wojciech, et al. “Charge-Carrier Transporting Graphene-Type Molecules†.” Chemistry of Materials, vol. 23, no. 3, Aug. 2011, pp. 554–567., doi:10.1021/cm102252w.
5. Warner, Jamie H., et al. Graphene: Fundamentals to emergent applications. Kidlington, Oxford, Elsevier, 2013.
6. Sharma, Kal R. Graphene nanomaterials. New York, Momentum Press, 2014.
7. Nazarpour, Soroush, and Stephen R Waite. Graphene technology: From Laboratory to Fabrication. Weinheim, Wiley Vch Publication, 2016.
8. Sood, Ashok K., et al. “Review of Graphene Technology and Its Applications for Electronic Devices.” Review of Graphene Technology and Its Applications for Electronic Devices | InTechOpen, InTech, 25 Nov. 2015. Accessed 20 Sept. 2017.